 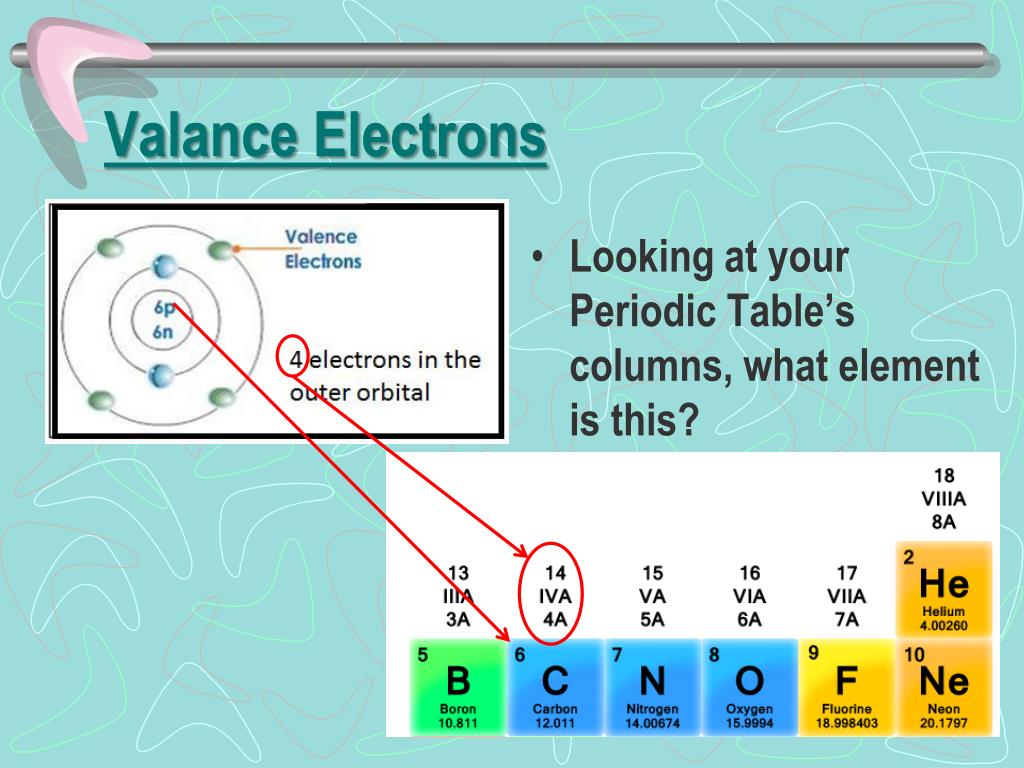
Periods are the horizontal rows of the periodic table, which run from 1 to 7 in number. In the periodic table, elements with chemical properties that are similar are grouped together in the same group. Groups are the vertical columns of the periodic table that are counted from left to right, from 1 to 18. Symbolised by the letter “O,” oxygen has an atomic number of eight and is represented by the letter “O.”ĭetermine the element’s group number and period number by referring to the periodic table. Each square on the periodic table has the letter symbol for an element printed directly below the atomic number of the element in the square in question.Ĭonsider the element oxygen, which can be found on the table. Locate the element you’re looking for on the periodic table. Finding Valence Electrons for All Elements Except Transition Metals However, although the orbitals involved can be found in an inner electron shell and do not all correspond to a certain electron shell or principal quantum number n in the same element as one another, they are all located at relatively close distances to the nucleus in the same element. The orbitals of the incomplete (n-1)d subshell are included for transition metals, and the orbitals of the incomplete (n-2)f and (n-1)d subshells are included for lanthanides and actinides. The valence shell of main-group elements is made up of the ns and np orbitals, which are located in the outermost electron shell. 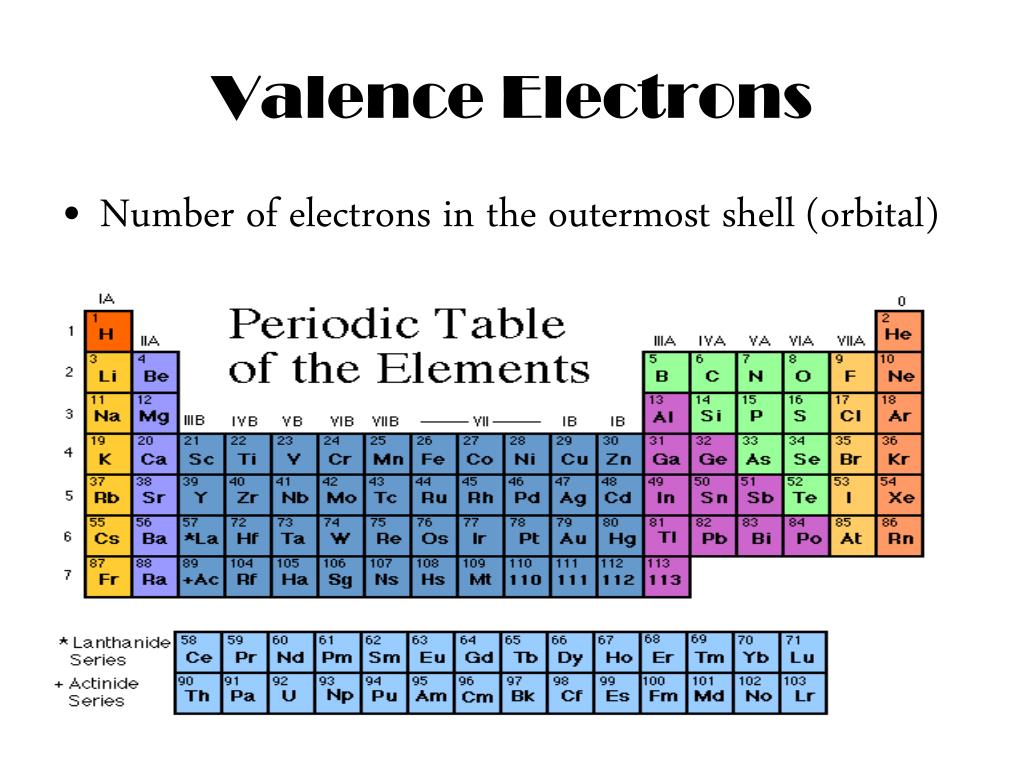 
As a result, valence electrons have a direct impact on the behaviour of elements during a chemical reaction. Atoms are more likely to accept or lose electrons if doing so will result in the completion of their outer shell. The valence electrons of an atom travel in the subshell that is the furthest away from the nucleus of the atom, according to its definition. Each electron shell is made up of one or more subshells, which are themselves composed of electrons. Electrons orbit around the atomic nuclei at specific energy levels, known as principal energy levels or electron shells, which are defined by the atom’s structure. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
